Ca element configuration8/12/2023  The chemistry of calcium is that of a typical heavy alkaline earth metal. 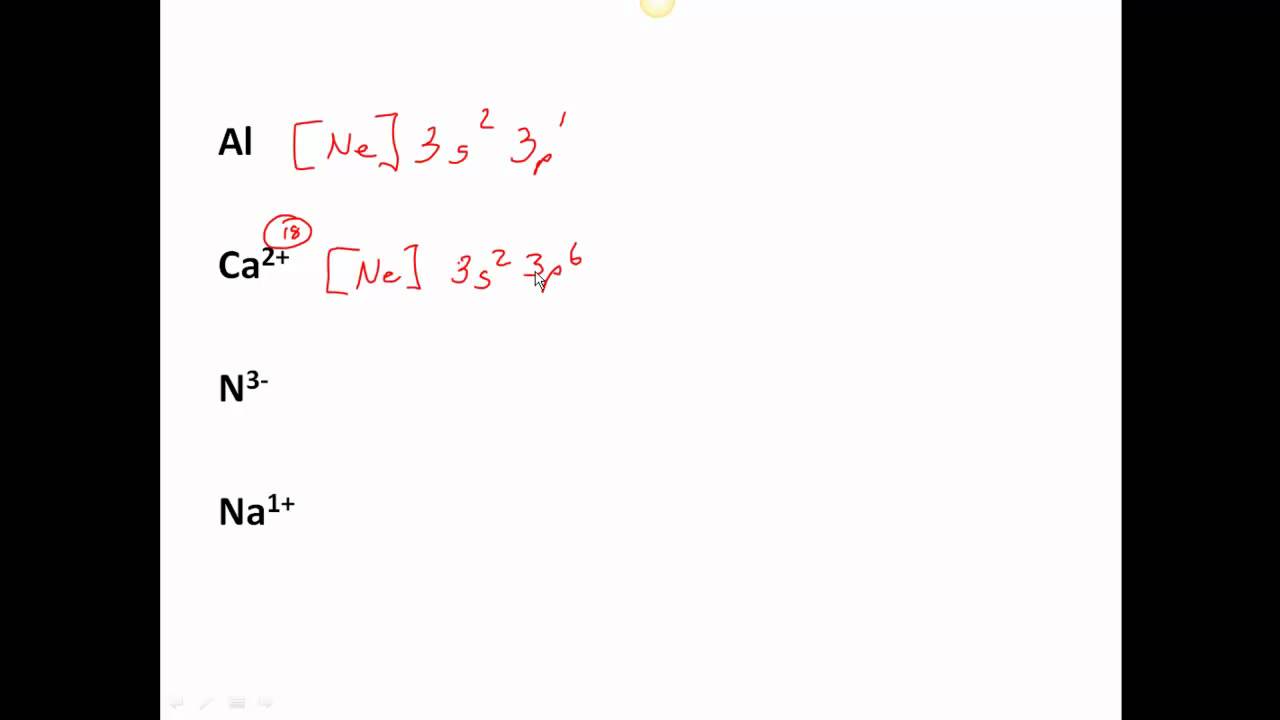
Structure of the polymeric 2+ center in hydrated calcium chloride, illustrating the high coordination number typical for calcium complexes. While calcium is infeasible as a conductor for most terrestrial applications as it reacts quickly with atmospheric oxygen, its use as such in space has been considered. While calcium is a poorer conductor of electricity than copper or aluminium by volume, it is a better conductor by mass than both due to its very low density. Ĭalcium is harder than lead but can be cut with a knife with effort. Its density of 1.55 g/cm 3 is the lowest in its group. It crystallises in the face-centered cubic arrangement like strontium above 450 ☌, it changes to an anisotropic hexagonal close-packed arrangement like magnesium. Physical propertiesĬalcium metal melts at 842 ☌ and boils at 1494 ☌ these values are higher than those for magnesium and strontium, the neighbouring group 2 metals. Nevertheless, beryllium and magnesium differ significantly from the other members of the group in their physical and chemical behaviour: they behave more like aluminium and zinc respectively and have some of the weaker metallic character of the post-transition metals, which is why the traditional definition of the term "alkaline earth metal" excludes them. Ĭalcium, strontium, barium, and radium are always considered to be alkaline earth metals the lighter beryllium and magnesium, also in group 2 of the periodic table, are often included as well. This occurs because of the much greater lattice energy afforded by the more highly charged Ca 2+ cation compared to the hypothetical Ca + cation. Hypothetical univalent salts of calcium would be stable with respect to their elements, but not to disproportionation to the divalent salts and calcium metal, because the enthalpy of formation of MX 2 is much higher than those of the hypothetical MX. Hence, calcium is almost always divalent in its compounds, which are usually ionic. Like the other elements placed in group 2 of the periodic table, calcium has two valence electrons in the outermost s-orbital, which are very easily lost in chemical reactions to form a dipositive ion with the stable electron configuration of a noble gas, in this case argon. A calcium atom has twenty electrons, arranged in the electron configuration 4s 2. Characteristics ClassificationĬalcium is a very ductile silvery metal (sometimes described as pale yellow) whose properties are very similar to the heavier elements in its group, strontium, barium, and radium. Calcium ions outside cells are important for maintaining the potential difference across excitable cell membranes, protein synthesis, and bone formation. As electrolytes, calcium ions (Ca 2+) play a vital role in the physiological and biochemical processes of organisms and cells: in signal transduction pathways where they act as a second messenger in neurotransmitter release from neurons in contraction of all muscle cell types as cofactors in many enzymes and in fertilization. On the other hand, the metal in pure form has few applications due to its high reactivity still, in small quantities it is often used as an alloying component in steelmaking, and sometimes, as a calcium–lead alloy, in making automotive batteries.Ĭalcium is the most abundant metal and the fifth-most abundant element in the human body. Calcium compounds are widely used in many industries: in foods and pharmaceuticals for calcium supplementation, in the paper industry as bleaches, as components in cement and electrical insulators, and in the manufacture of soaps. Pure calcium was isolated in 1808 via electrolysis of its oxide by Humphry Davy, who named the element. Some calcium compounds were known to the ancients, though their chemistry was unknown until the seventeenth century. The name derives from Latin calx " lime", which was obtained from heating limestone. The most common calcium compound on Earth is calcium carbonate, found in limestone and the fossilised remnants of early sea life gypsum, anhydrite, fluorite, and apatite are also sources of calcium. It is the fifth most abundant element in Earth's crust, and the third most abundant metal, after iron and aluminium. 
Its physical and chemical properties are most similar to its heavier homologues strontium and barium. As an alkaline earth metal, calcium is a reactive metal that forms a dark oxide-nitride layer when exposed to air. 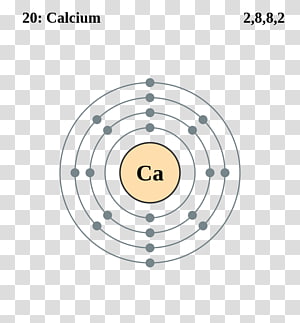
Calcium is a chemical element with the symbol Ca and atomic number 20. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
